 
Valence shell electron-pair repulsion theory (VSEPR theory) enables us to predict the molecular structure, including approximate bond angles around a central atom, of a molecule from an examination of the number of bonds and lone electron pairs in its Lewis structure. These examples reveal that two to six bonding electron groups around the central atom lead to five basic molecular shapes: linear, trigonal planar, tetrahedral, trigonal bipyramidal, and octahedral. All the bonds are equivalent and bond angles are 90°. The geometry of the molecule is octahedral. The two other groups lie on either side of this plane. In sulfur hexafluoride, there are six electron groups around the sulfur atom. The molecule has trigonal bipyramidal geometry. The angle between the equatorial and axial chlorines is 90°. There is one chlorine atom each above and below the plane. The three equatorial chlorine atoms are separated by the bond angle of a 120° and assume a trigonal planar arrangement. Phosphorus pentachloride has five electron groups around the central atom. If five balloons are tied together, maximum separation is achieved when the three balloons are in one plane, and the remaining two are placed on either side of the plane. They are farthest when the bond angle is 109.5°, and the molecule assumes a three-dimensional tetrahedral geometry. In the case of methane, there are four electron groups surrounding the central carbon atom. 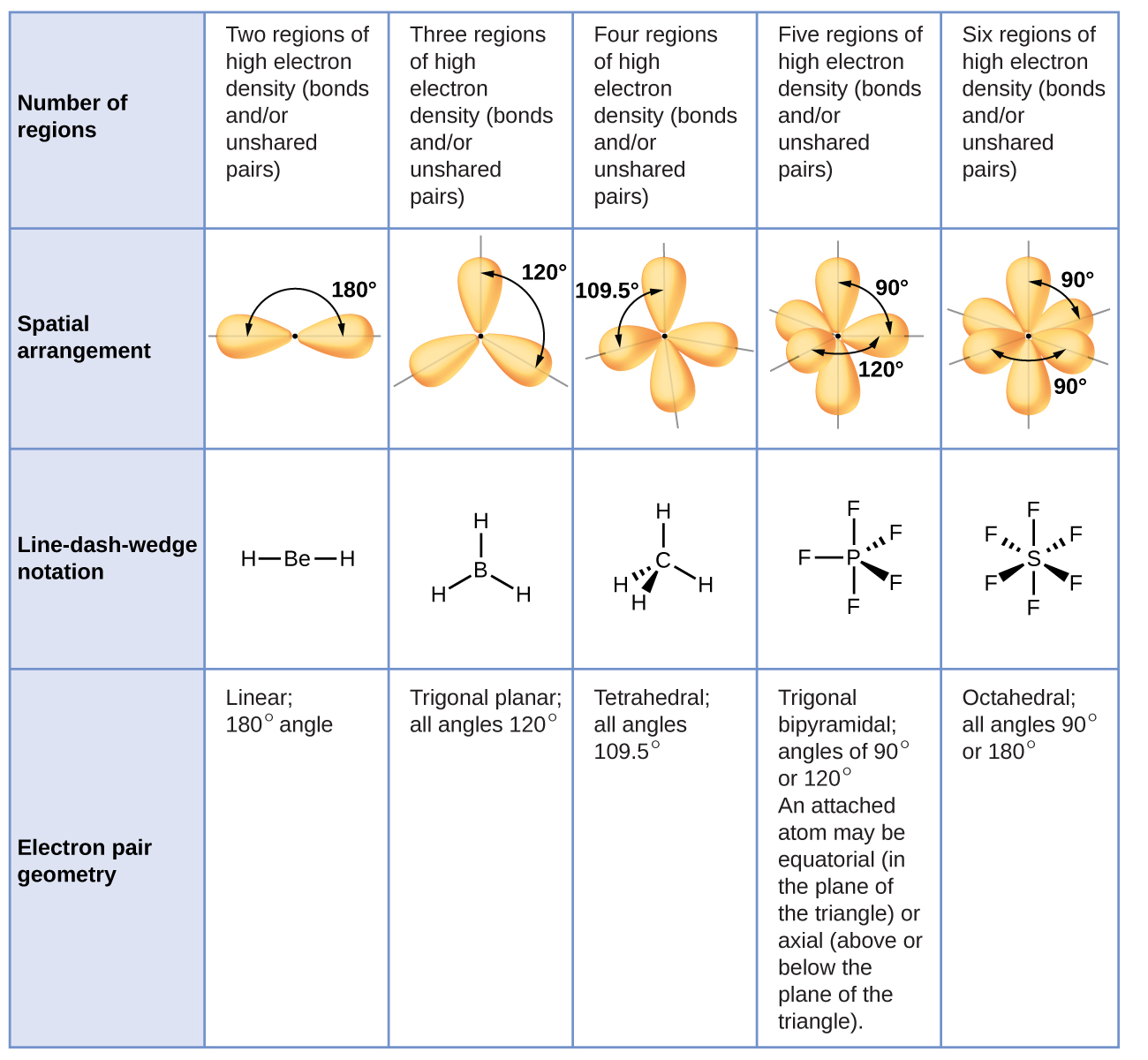
VSEPR theory predicts that the molecule exhibits trigonal planar geometry. The repulsion between these groups can be minimized by assuming a 120° bond angle. Thus, the bond angle is 180°, and the molecular shape is linear.īoron trifluoride has three electron groups around the central boron atom. According to VSEPR, the minimum repulsion between these electron groups is achieved through a maximum separation. Molecular geometry is dictated by the arrangement of various electron groups around the central atom.īeryllium fluoride has two electron groups around the central atom. Each balloon orients itself away from the other as much as possible. It assumes that the negatively charged electron groups, which may be electrons involved in a single bond, multiple bonds, or lone pairs, repel one another and try to stay at the maximum possible distance from each other to minimize repulsions. Valence shell electron-pair repulsion or VSEPR theory serves as a tool to predict molecular structure. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
